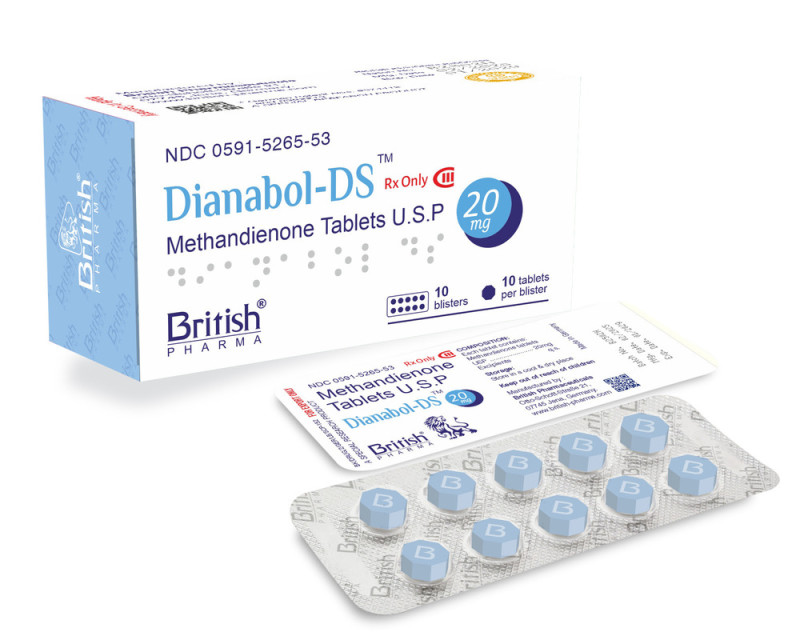
Dian Abol Ds 20mg
Dian Abol DS 20 mg is an oral anabolic formulation containing methandrostenolone, a synthetic anabolic androgenic compound known for its rapid anabolic activity. It is designed to support increased protein synthesis and positive nitrogen balance, whiRead More
-
Active Ingredient:
Methandrostenolone
-
Dosage Strength:
20 Mg Per Tablet
-
Drug Category:
Anabolic-androgenic Steroid
-
Chemical Formula:
C20H28O2