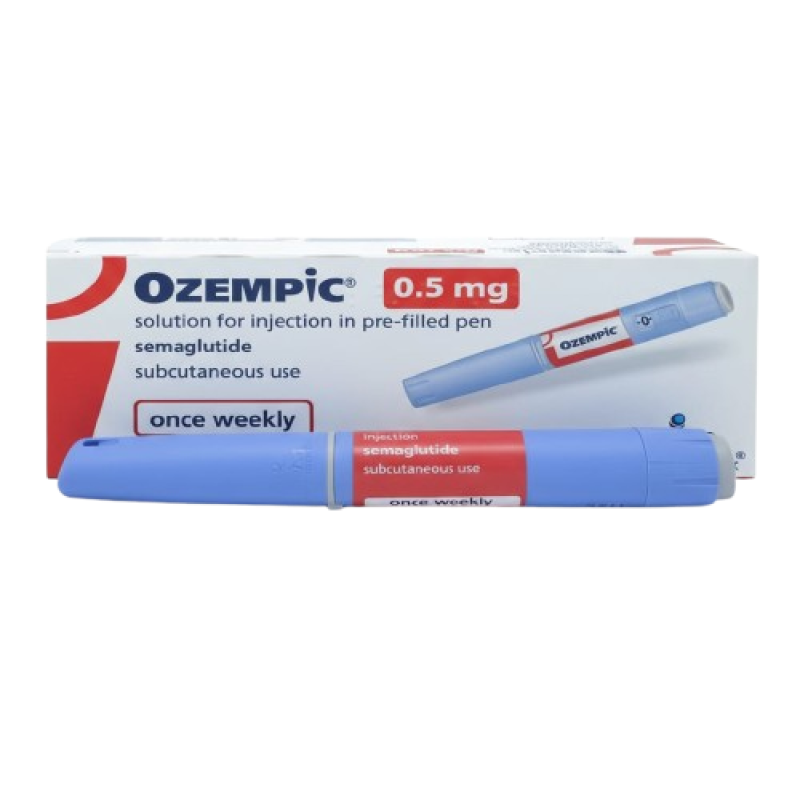
Ozempic 0.5 Mg Injection
Ozempic 0.5 mg Injection is a prescription injectable medicine containing semaglutide, a glucagon-like peptide-1 (GLP-1) receptor agonist. It is designed to support blood glucose control by enhancing glucose-dependent insulin secretion, reducing exceRead More
-
Active Ingredient:
Semaglutide
-
Dosage Strength:
0.5 Mg
-
Drug Class:
GLP-1 Receptor Agonist
-
Product Type:
Injectable Medication