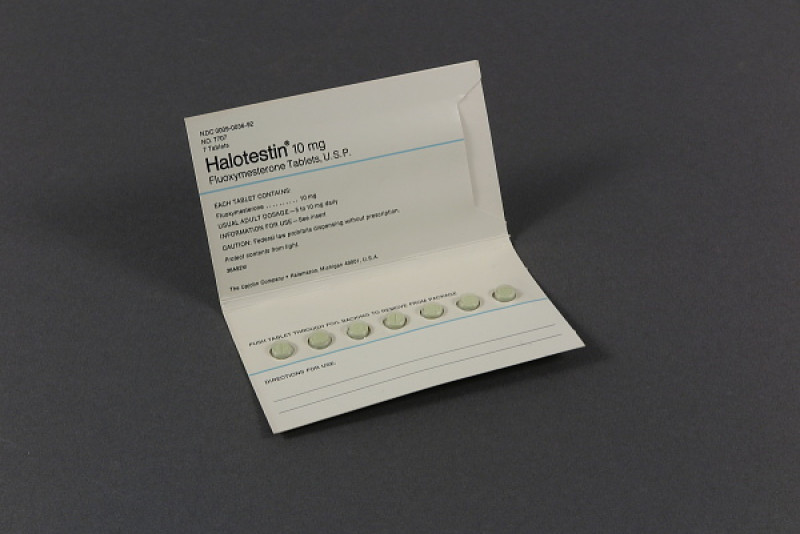
Halotestin (fluoxymesterone) Tablets 10mg
Halotestin (Fluoxymesterone) Tablets 10 Mg are an oral anabolic androgenic medication containing fluoxymesterone, a synthetic androgen derived from testosterone. It is known for its strong androgenic activity and is used in specific medical settings Read More
-
Ingredient:
Fluoxymesterone
-
Dosage Strength:
10 Mg Per Tablet
-
Drug Category:
Anabolic Androgenic Steroid
-
Route Of Administration:
Oral