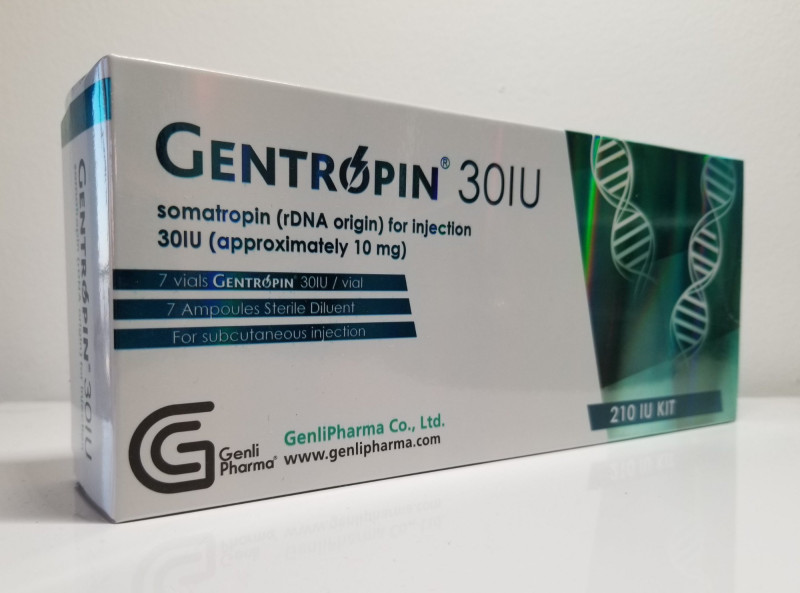
Gentropin 30iu
Gentropin 30 IU is a recombinant human growth hormone (somatropin) formulation developed to mirror the natural growth hormone produced by the human pituitary gland. It is used in clinically approved settings to support normal growth, cell regeneratioRead More
-
Hormone Content:
30 IU
-
Generic Name:
Somatropin
-
Drug Class:
Growth Hormone
-
Dosage Form:
Injectable Peptide