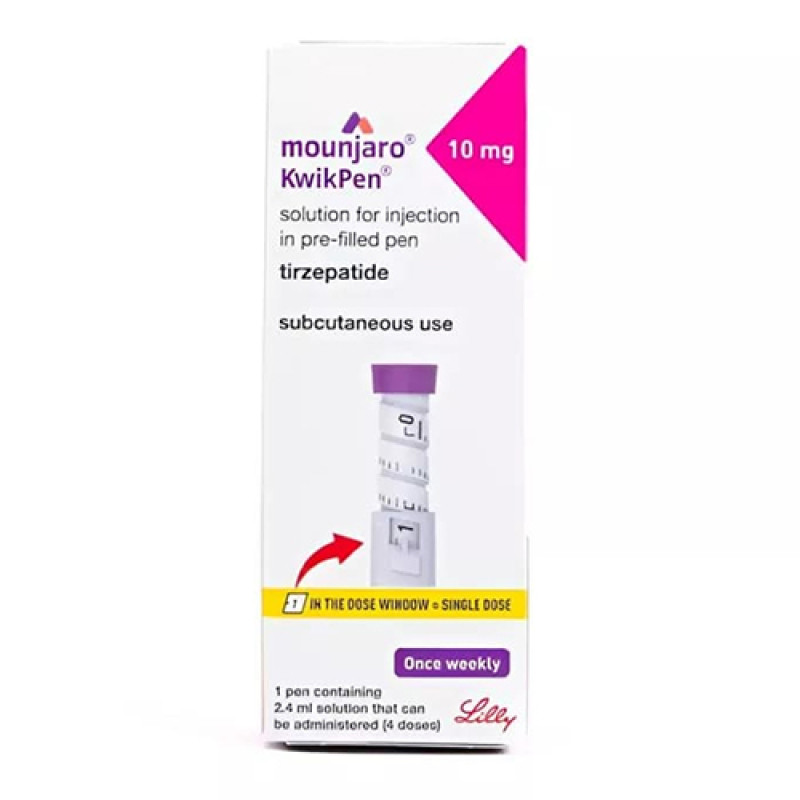
Tirzepatide 10 Mg Injection
Tirzepatide 10 Mg Injection is a long-acting injectable formulation developed for metabolic and endocrine management. It is a dual-acting peptide that targets both glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1)Read More
-
Dosage Strength:
10 Mg
-
Drug Class:
Dual GIP / GLP-1 Receptor Agonist
-
Active Ingredient:
Tirzepatide
-
Product Category:
Antidiabetic Medication